Unleash the power of AI for developers by improving processes across your software delivery organization
Unleash the power of AI for developers by improving processes across your software delivery organization
Trusted by Enterprise Customers
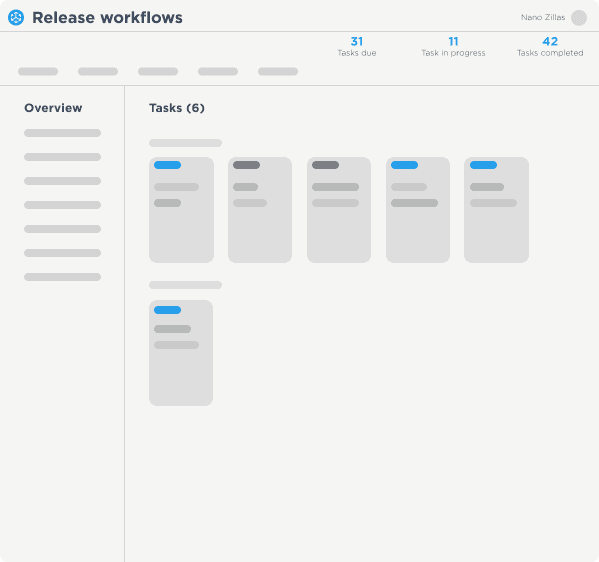
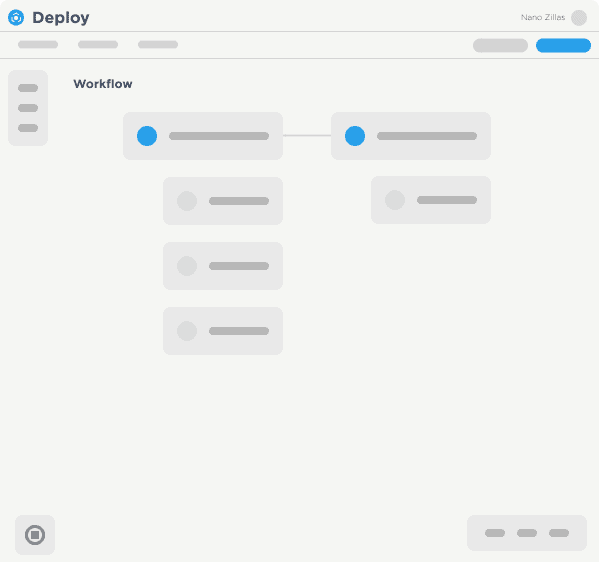
Solutions That Manage The Complexities Of Software Delivery
The World’s Largest Enterprises Depend on Digital.ai to Automate Software Releases
%
of Fortune 100 Companies
 57Worldwide Government Entities
57Worldwide Government Entities
 57Worldwide Government Entities
57Worldwide Government Entities
Explore Products Available On Our AI-Powered DevSecOps Platform
Our products help the world’s largest organizations automate software releases, improve mobile application testing and security, and provide insights across the software lifecycle.
How to Make AI-generated code Secure, Private and Compliant

EXPLORE
Related Resources

See our solutions in action
Our team of experts are available to help accelerate your digital transformation